In a research proposal for sumanene chemistry prepared in June 1998, H. Sakurai outlined a synthetic route to sumanene.
Source of Scheme: H. Sakurai, Bull. Chem. Soc. Jpn. 2021, 94, 1579-1587
In the intricate world of chemistry, certain compounds stand out not only for their intriguing structures but also for the potential they hold for various applications. One such compound is sumanene, a bowl-shaped polycyclic aromatic hydrocarbon that has captured the interest of scientific attention as it resembles a part of buckminsterfullerene.
At the heart of sumanene lies a benzene ring, a fundamental building block in organic chemistry. Surrounding this central ring are alternating benzene and cyclopentadiene rings, creating a unique polycyclic structure. A groundbreaking method for crafting this exquisite molecule was detailed in the esteemed journal Science (Science 2003, 301, 1878). Following the disclosure of its synthetic blueprint, numerous scientists embarked on investigating sumanene to unlock its myriad possibilities.
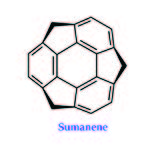
The structure of sumanene.
Let’s delve into the successful chemical synthesis method of sumanene. Before its publication in the journal Science, numerous chemists struggled to identify a viable synthetic route. How did H. Sakurai manage to pioneer an intriguing chemical synthesis pathway for this unique molecular architecture? This question piqued the interest of the editor, who sought to uncover Sakurai’s journey to success. Fortunately, an article appeared in the Bulletin of the Chemical Society of Japan (https://doi.org/10.1246/bcsj.20210046), where Professor Sakurai shared his narrative on establishing himself in sumanene and azasumanene chemistry from 1993 to 2014.
In late October 1993, Sakurai found himself engrossed in the Chemistry Department library at the University of Tokyo. His attention was captured by a manuscript from Prof. Mehta’s group in JCS Chem. Commun. What struck Sakurai was the tone of the paper—it seemed to convey a sense of “failure” regarding the synthesis of sumanene. He couldn’t fathom why even Prof. Mehta had encountered difficulties in synthesizing sumanene. This moment marked the inception of Sakurai’s journey into sumanene chemistry, igniting his fascination with the π-figuration concept. The editor was impressed by how Sakurai found inspiration from what seemed like a failure.
He initiated a retrosynthetic analysis of sumanene in his notebook, all the while concentrating on organometallic chemistry. Three years later, during his tenure as a JSPS postdoc in Prof. Chuck Casey’s group at the University of Wisconsin-Madison (1996-1998), he concentrated on enhancing experimental methods and delving into organometallic chemistry. This period granted him the liberty to ponder upcoming projects, including the synthesis of sumanene.
Scheme depicting the first retrosynthesis of sumanene, as drawn in
Sakurai’s notebook in October 1993.
Source of Scheme: H. Sakurai, Bull. Chem. Soc. Jpn. 2021, 94, 1579-1587
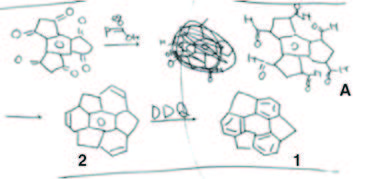
In October 1996, he devised a second retrosynthesis plan, identifying the cyclotrimerization of norbornadiene as a promising precursor for sumanene synthesis. This plan involved converting norbornadiene to sumanene through ozonolysis, pinacol coupling, and dehydrative aromatization.
Sakurai outlined the second retrosynthetic approach for sumanene (1) in his notebook during October 1996.
Source of Scheme: H. Sakurai, Bull. Chem. Soc. Jpn. 2021, 94, 1579-1587
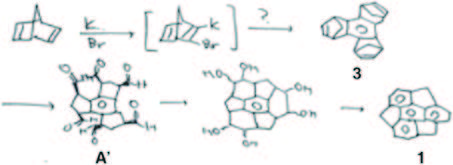
Soon after attending a lecture by Prof. Grubbs, he conceived by De Lucchi’s group. Given the undesired formation of the anti isomer as the predominant product and the potential challenge of separation, his initial focus was on developing a new method for achieving syn-selective cyclotrimerization. Despite nearly two years of effort, tangible results remained elusive. The trials were continued after he moved to Osaka University (from April 2000). After numerous failed attempts, he finally achieved his desired molecular structure, sumanene, by the idea of tandem metathesis. Following this presentation, he sketched out a reaction layout in his notebook. Feeling confident that he had gathered all the necessary components for synthesizing sumanene, he made the decision to commence sumanene chemistry upon returning to the University of Tokyo as an assistant professor in April 1998.
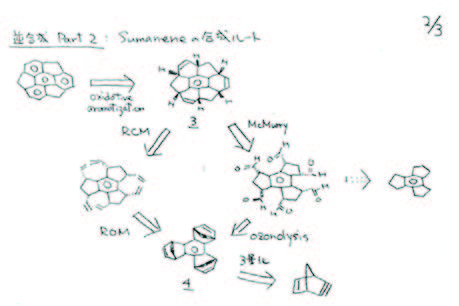
In order to defend his idea of sumanene synthesis at the University of Tokyo, he primarily considered two approaches: one involving ozonolysis and the McMurry reaction, and the other employing tandem ring-opening metathesis (ROM) and ring-closing metathesis (RCM).
He commenced the research with the cyclotrimerization of the norbornene unit, adopting the method employed by De Lucchi’s group. Given the undesired formation of the anti isomer as the predominant product and the potential challenge of separation, his initial focus was on developing a new method for achieving syn-selective cyclotrimerization. Despite nearly two years of effort, tangible results remained elusive. The trials were continued after he moved to Osaka University (from April 2000). After numerous failed attempts, he finally achieved his desired molecular structure, sumanene, by following scheme 1. Confirmation was provided by 1H NMR, which displayed only one singlet peak and doublet pairs, strongly suggesting the formation of sumanene (1).
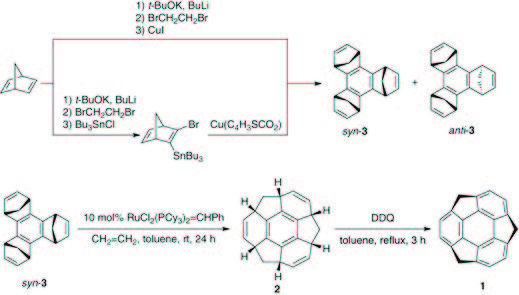
Scheme 1. The synthetic pathway to sumanene (1) led H. Sakurai to achieve a distinct 1H-NMR signal for product.
Source of Scheme: H. Sakurai, Bull. Chem. Soc. Jpn. 2021, 94, 1579-1587
Even after successfully synthesizing sumanene, his ultimate dream of sharing it with the scientific community remained unfulfilled until he could publish his manuscript. Initially submitted to the Journal of the American Chemical Society, it faced rejection due to insufficient evidence for compound 1, lacking elemental analysis or a crystal structure. To expedite the process, he submitted the manuscript to Science while continuing to gather data. Unexpectedly, he received correspondence from Science’s editorial office, and his manuscript was accepted for publication. This journey from aspiration to accomplishment in the chemical synthesis of sumanene, dream initiated in 1993, finally became a reality in 2003.

Professor Hidehiro Sakurai, Osaka University, Japan
Hidehiro Sakurai earned his Ph.D. from The University of Tokyo in 1994, studying under Professor Koichi Narasaka. Following this, he served as an Assistant Professor in Tokyo under Professor Narasaka’s guidance and later as a JSPS Postdoctoral fellow at the University of Wisconsin–Madison with Professor C. P. Casey. In 2000, he joined Osaka University, transitioning to the Institute for Molecular Science (IMS) in 2004. Returning to Osaka University in 2014, he now holds the position of Professor at the Division of Applied Chemistry. His current research focuses on buckybowls, nonplanar π-aromatic compounds, and nanocatalyst development.
– RN Dhital,
PhD Editor-in-Chief