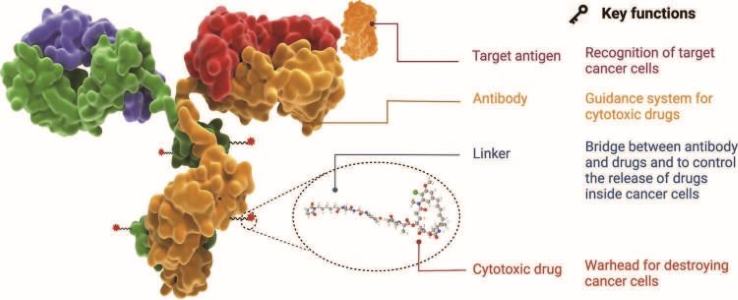
The structure of a typical ADC drug.
Photo Source: Zhang et al., Sig Transduct Target Ther 2022, 7, 93
Antibody Drug Conjugates (ADCs), a new modality for targeted cancer therapy has received significant attention in recent years. A typical ADC is composed of three key elements; a monoclonal antibody (mAb) covalently attached to a cytotoxic drug (payload) via a chemical linker. ADCs benefit from the combination of highly specific targeting ability of antibody and highly potent killing effect of a payload to achieve effective elimination of cancer cells while sparing healthy cells. Due to its ability to target specific proteins on the surface of cancer cells, ADC has revolutionized the landscape for cancer treatment by delivering toxic payload selectively to the tumors resulting an enhanced efficacy compared to traditional chemotherapy.
Mechanism of Action:
Once the ADC reaches to target cell, ADC complex induces apoptosis in cancer cells in several stages: First, ADC binds to surface of cancer cells forming ADC–antigen complex (cell surface binding). ADC–antigen complex is then internalized within the cancer cell by endocytosis to form an early endosome, which after maturation, fuses with lysosomes releasing cytotoxic payload which eventually causes the cell death or apoptosis.
The concept of targeted cancer therapy/ADC is not new. Paul Ehrlich, a Nobel laureate in medicine, envisioned if a substance is capable of binding selectively to a cancer cell, then drug can be delivered to the cancer cells via targeted drug delivery in 1910, famously known as “magic bullet”. Ninety years later, after scientific and technological advancements, the first ADC drug Mylotarg® (gemtuzumab ozogamicin) approved by the US Food and Drug Administration (FDA) for the treatment of acute myeloid leukemia in 2000. Eleven years later, second ADC drug Adcetris® (brentuximab vedotin) was approved for the treatment of Hodgkin lymphoma. After that, subsequent developments in the technology led to rapid growth in regulatory approvals for ADCs targeting variety of tumor types. Currently, 14 ADCs has already received market approval worldwide and over 100 ADCs are at different stages of clinical development.
– Buddha Khatri, PhD
Scientist, Pharmaceutical Company, USA